Abstract
Background
Bortezomib with lenalidomide and dexamethasone (VRd) is a standard regimen for the induction treatment of multiple myeloma (MM) transplant eligible and ineligible patients. Deregulation of histone acetylation has been recognized to serve a critical role in the pathogenesis of MM, which histone deacetylase (HDACs) are overexpressed in plasma cells derived from patients with MM. Therefore, HDACs may promise targets for MM therapy, and panobinostat was approved by FDA for the treatment of relapsed/refractory MM in 2015. Chidamide, a novel oral benzamide type HDAC inhibitor independently developed and approved as anticancer drugs in China, selectively suppresses the activity of class I HDACs. Our studies have shown that chidamide can enhance cytotoxic effect of bortezomib or lenalidomide on MM cells in vitro, which suggested a synergistic combination of chidamide with bortezomib or lenalidomide at low dose. In ASH 2019 we reported Phase 1 Suzhou MM02 study in which chidamide at 4 dose levels (15mg/20mg/25mg/30mg d 0,3,7,10) were administered to 12 patients with VRd for 4-cycle induction therapy. No DLT in Chi-VRd group were observed. This phase 2 trial was designed to further assess the safety and efficacy of Chi-VRd versus VRd for induction treatment in patients with newly diagnosed high risk MM before autologous transplantation (NCT 04025450).
Methods
In this study, we enrolled patients from the First Affiliated Hospital of Soochow University and Soochow Hopes Hematology Hospital with newly diagnosed high-risk multiple myeloma who were aged 18 years or older and eligible for autologous stem-cell transplant (ASCT) according to International Myeloma Working Group diagnostic criteria. All the patients were randomly assigned to Chi-VRd and VRd group. Chi-VRd group patients received Chidamide 20mg orally on days -1,2,6 and 9 in combination with VRd (bortezomib 1.3mg/m2 subcutaneously on days 1,4,8,11; lenalidomide 25mg orally on days 1-14; dexamethasone 20mg iv or orally on days 1,2,4,5,8,9,11,12) for 4 cycles. VRd group patients received VRd (bortezomib 1.3mg/m2 subcutaneously on days 1,4,8,11; lenalidomide 25mg orally on days 1-14; dexamethasone 20mg iv or orally on days 1,2,4,5,8,9,11,12) for 4 cycles.
All the patients proceed with ASCT after four cycles or continue VRd therapy for up to eight cycles, followed by maintenance therapy for up to 2 years. Primary efficacy endpoints were overall response rate (ORR), rate of very good partial response (VGPR) or better and rate of complete response (CR) for Chi-VRd and VRd. Safety data described rates of adverse events.
Results
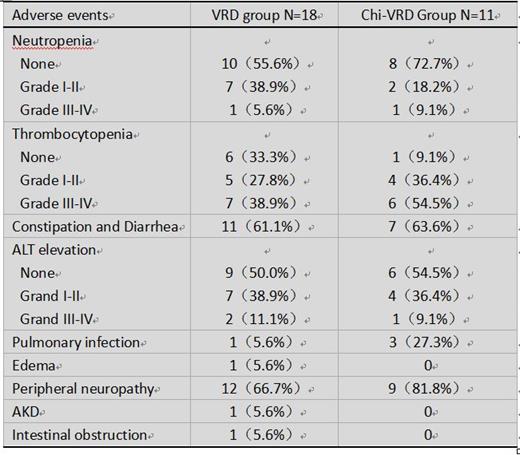
Between Mar 16, 2020, and Jul 22, 2021, 40 patients were enrolled and randomly assigned to either the VRd regimen (n=20) or the Chi-VRd regimen (n=20). In the Chi-VRd group, 1 patient was discharged due to severe tumor lysis syndrome, and another 1 patient was discharged from sick sinus syndrome (considering lenalidomide related). By the deadline for submission, 18 patients in the VRd group and 11 patients in the Chi-VRd group had completed induction chemotherapy. The total adverse events were listed in the table below. The main treatment-related adverse events were hematological toxicity, hepatotoxicity, constipation, and peripheral neuropathy. ORR was 90.9% (10/11) for Chi-VRd and was 100% for VRd. Response of VGPR or better was 81.8% (9/11) for Chi-VRd and was 83.3% (15/18) for VRd. And rate of CR was 63.6%(7/11) for Chi-VRd and was 44.4%(8/18) for VRd.
Conclusion
The Chi-VRd group had more adverse events of thrombocytopenia, peripheral neuropathy and pulmonary. No treatment-related death was observed in two groups. Patients received Chi-VRd get more CR rate. Updated PFS and OS with comparison between VRd and Chi-VRd will be presented at the following study.
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal